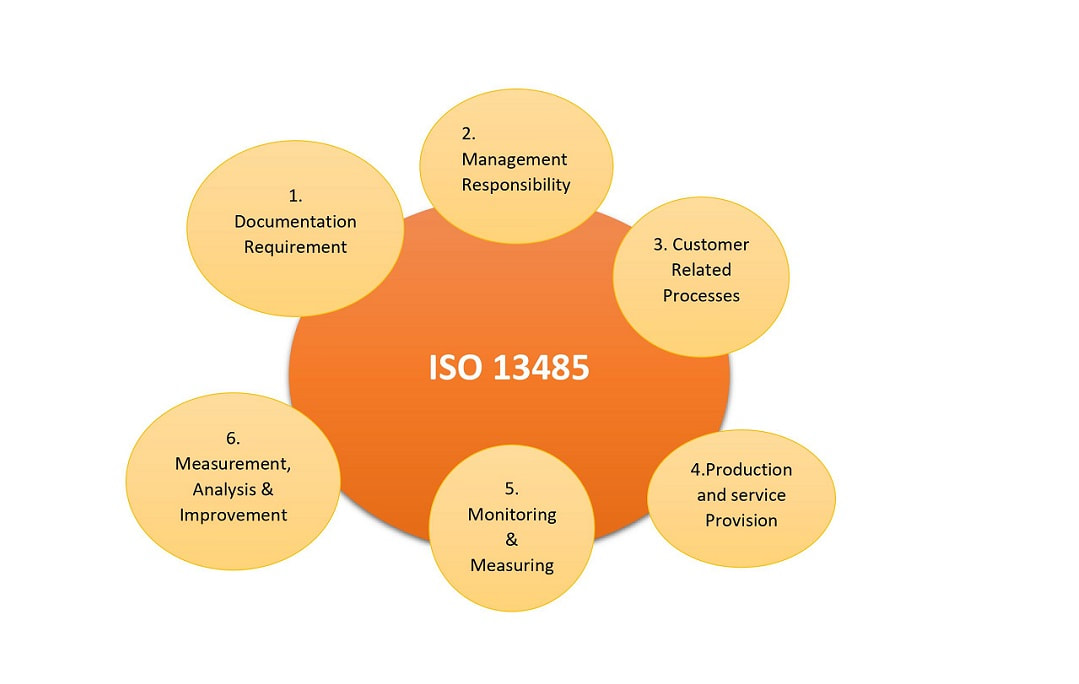
 ISO 13485: ISO 13485 is quality management system for medical devices commonly termed as Medical Devices Quality Management System. The ISO 13485 standard requirements are the foundation upon which a robust quality management system can be built. The implementation of QMS through ISO 13485 enables your organization to identify risks relating to possible non-conformances and reduce or prevent the chances of expensive and highly damaging accidents. It also allows to enhance your public image, increase your brand equity and improve your market share. The implementation of 13485 is dependent on the following parts of the standard:
ISO 13485 Certification requires commitment and effort. This increased efficiency adds to the credibility of an organization with potential customers. It is through these core principles that medical device manufacturers can successfully implement the requirements outlined in the QMS. Requirement:
Importance of Certificate & Consultants: Achieving Certificate allows you to expand your business to the maximum level. It helps you to increase bottom line of your organization and ensure FDA Compliance. Our consultants makes sure that your organization achieve certificate by ensuring that the medical devices being manufactured are up to mark, follow every quality aspect, and are safest to use. Our ISO 13485 consulting and project management teams work closely with you to implement your QMS on time and on budget. Certification can also expand your market access and improve your company's profitability.
Glenview Services: Our company provides highly skilled ISO 13485 Consultants to assist you obtain Certificate and guide you for better understanding of Standards. For more information visit our website: https://glenviewgroup.com/iso-13485-consulting/.
0 Comments
 ISO 27001 does not formally mandate specific information security controls since the controls that are required vary markedly across the wide range of organizations adopting the standard. The information security controls from ISO27002 are summarized in annex A to ISO27001, rather like a menu. Organizations adopting ISO 27001 are free to choose whichever specific information security controls are applicable to their particular information risks, drawing on those listed in the menu and potentially supplementing them with other a la carte options (sometimes known as extended control sets). As with ISO 27002, the key to selecting applicable controls is to undertake a comprehensive assessment of the organization’s information risks, which is one vital part of the ISMS. Furthermore, management may elect to avoid, share or accept information risks rather than mitigate them through controls - a risk treatment decision within the risk management process. Annex A Reference control objectives and controls - little more than a list of titles of the control sections in ISO 27002. The annex is ‘normative’, implying that certified organizations are expected to use it, but the main body says they are free to deviate from or supplement it in order to address their particular information risks. Annex A alone is hard to interpret. Please refer to ISO 27002 for more useful detail on the controls, including implementation guidance. The Information Security Controls under ISO 27001 are grouped as under:
 What is ISO 9001? ISO 9001 is a Quality Management System standard designed to focus on quality services/products and customer satisfaction. Why ISO 9001 Certification and Auditor Training? ISO 9001 belongs to the family of ISO 9000 standards and is the only standard in the family that requires certification. This certification is a great help for organizations to meet their customer requirements while fulfilling any regulatory requirements. An unproductive audit can mean extreme consequences which results in customer dissatisfaction, process failure, and regulatory noncompliance. ISO 9001 certification and audit training course helps delegates in optimizing their auditing skills with the internationally recognized ISO 9001 standard and boost their internal audit capabilities. Our experts help you in gaining confidence in planning and performing an effective audit. Further, they ensure that you get necessary skills related to reporting and assessing corrective action where necessary. Delegates planning to become Lead Auditors can also meet their requirements by attending our ISO 9001 Lead Auditor Course. Those managers and executive who want to implement ISO 9001 in their organization for certification can benefit by attending our ISO 9001 Certification Courses. What will you learn? After completing your ISO 9001 Certification And Auditor Training course, you will understand:
Prerequisites Delegates taking this course should have prior knowledge of:
GlenView Group, Inc. has years of experience in ISO courses. Our courses are tailored and delivered by the experts of the industry who support you on your professional journey. For further information please contact us and book your place for ISO 9001 certification and audit training courses. Introduction:
ISO 9001 is a quality management system standard, that focuses on the quality of the product or service an organization is providing, by improving the management of the organization in many relevant areas. Badly designed and applied Quality Management Systems (QMS) can cause serious detrimental burdens on an organization, making it very difficult to keep customers satisfied and attain business goals. A developed and properly implemented quality management system has its positive and long-term footprints, which include;
GlenView Group can help your organization in the development and implementation of the QMS to assure its immediate and long-term success. Our experts will assess, design and assist in developing and implementing your QMS. At GlenView, we believe in applying best practices gained from experience in multiple industries to provide a well-balanced QMS solution for our customers. Benefits of ISO 9001 Consulting: It is possible for an organization to develop and implement a Quality Management System without any help. However, an unplanned and unguided QMS development and implementation plan usually produces suboptimal results. The QMS Consulting experts at GlenView Group benefits your company by quickly applying our QMS knowledge to your unique circumstances. Our staff has acquired their QMS knowledge by being at the forefront of management system best practices, specifically Management System Support. Benefits of QMS Consulting include:
GlenView Group Inc. is an accredited training organization and offers public as well as online consulting classes. Our experts customize courses when we provide it on-site so that it makes sense to the people and is relevant and meaningful to the organization’s needs. |
|


