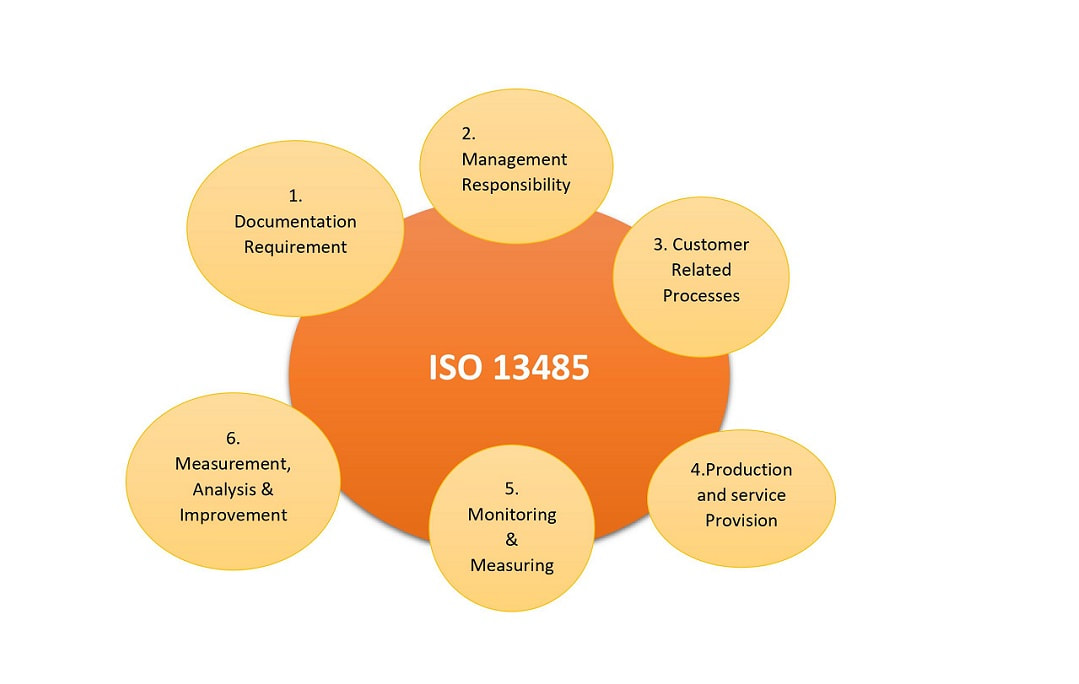
 ISO 13485: ISO 13485 is quality management system for medical devices commonly termed as Medical Devices Quality Management System. The ISO 13485 standard requirements are the foundation upon which a robust quality management system can be built. The implementation of QMS through ISO 13485 enables your organization to identify risks relating to possible non-conformances and reduce or prevent the chances of expensive and highly damaging accidents. It also allows to enhance your public image, increase your brand equity and improve your market share. The implementation of 13485 is dependent on the following parts of the standard:
ISO 13485 Certification requires commitment and effort. This increased efficiency adds to the credibility of an organization with potential customers. It is through these core principles that medical device manufacturers can successfully implement the requirements outlined in the QMS. Requirement:
Importance of Certificate & Consultants: Achieving Certificate allows you to expand your business to the maximum level. It helps you to increase bottom line of your organization and ensure FDA Compliance. Our consultants makes sure that your organization achieve certificate by ensuring that the medical devices being manufactured are up to mark, follow every quality aspect, and are safest to use. Our ISO 13485 consulting and project management teams work closely with you to implement your QMS on time and on budget. Certification can also expand your market access and improve your company's profitability.
Glenview Services: Our company provides highly skilled ISO 13485 Consultants to assist you obtain Certificate and guide you for better understanding of Standards. For more information visit our website: https://glenviewgroup.com/iso-13485-consulting/.
0 Comments
Leave a Reply. |
|

